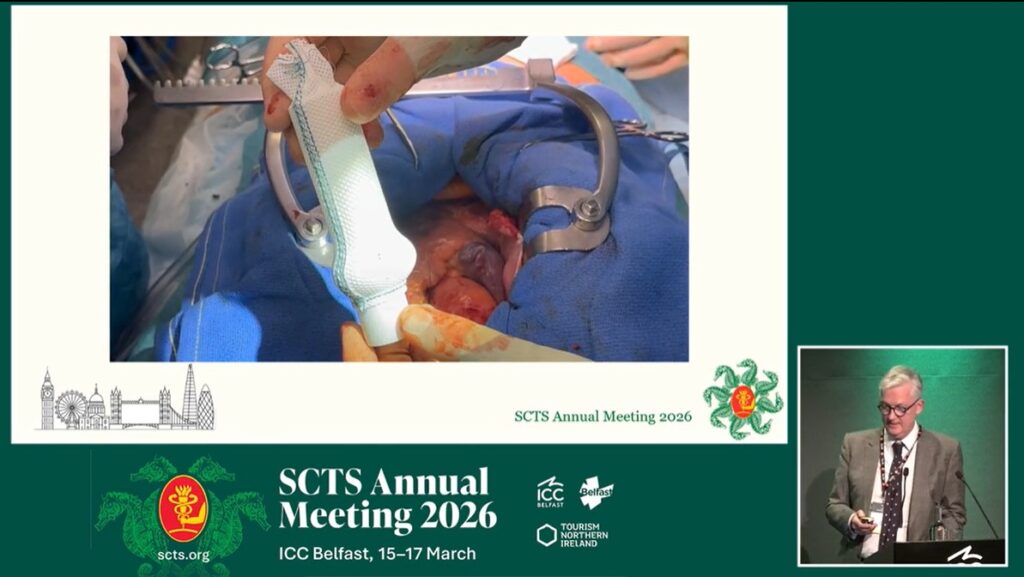
Following the completion of the initial clinical trial the Clinical Practice Committee at the Royal Brompton Hospital has approved the device for use in the treatment of suitable patients with a dilated aorta due to Marfan syndrome.
ExoVasc® PEARS
Menu